|
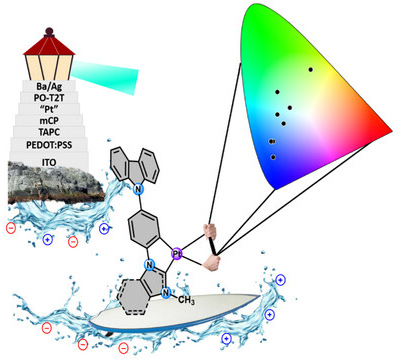
This paper describes the stage-wise synthesis of the new carbazole-appended platinum (II) cyclometalated N-heterocyclic carbene complex, [Pt(Cbz-C^C*bzim)Cl(NCCH3)] (A) (HC^C* = 1-(4-(9H-carbazol-9-yl)phenyl)-3-methyl-1H-benzimidazol-2-ylidene) from 9-(4-bromo-phenyl)-9H-carbazole, benzimidazole and [{Pt(μ-Cl)(η3-2-Me-C3H4)}2]. Complex A, and the analogous imidazole derivative [Pt(Cbz-C^C*im)Cl(NCCH3)] (B), are the precursors of the neutral complexes [Pt(Cbz-C^C*bzim/im)(acac)] (1A/B, acac: acetylacetonate), [Pt(Cbz-C^C*bzim/im)(P^O1)] (2A/B, P^O1: 2-diphenylphosphinobenzoate) and [Pt(Cbz-C^C*bzim/im)(P^O2)] (3A/B, P^O2: 2-diphenylphosphinophenolate). At low doping concentration in poly(methylmethacrylate (PMMA) films (1% for 1A/B and 5% for 2A/B and 3A/B), they show a bright phosphorescent emission, peaking in the blue (1B-3B) or blue–green (1A-3A) spectral range. The planar structure of 1A and 1B enables intermolecular interactions, especially for 1A, with a more extended π–system, which changes the color from blue to yellowish–green at higher doping concentration. Organic Light-Emitting Diodes (OLEDs) using 1A and 1B as emitters in thick doped and in ultrathin non-doped emitting layers show aggregate-derived emission even at very low concentrations. The best performing devices in this work use 1A and 3A and show a turn-on voltage of 2.99 and 2.55 V, a peak luminance of 39208 and 25837 cd m-2, and a peak current efficiency of 44.9 and 40.0 cd A-1 (12.8% and 13.4% EQE).
|